If you're diving into the exciting world of electrochemistry, biochemistry, sensor development, or particularly, battery research, knowing the ins and outs of a potentiostat is indispensable.
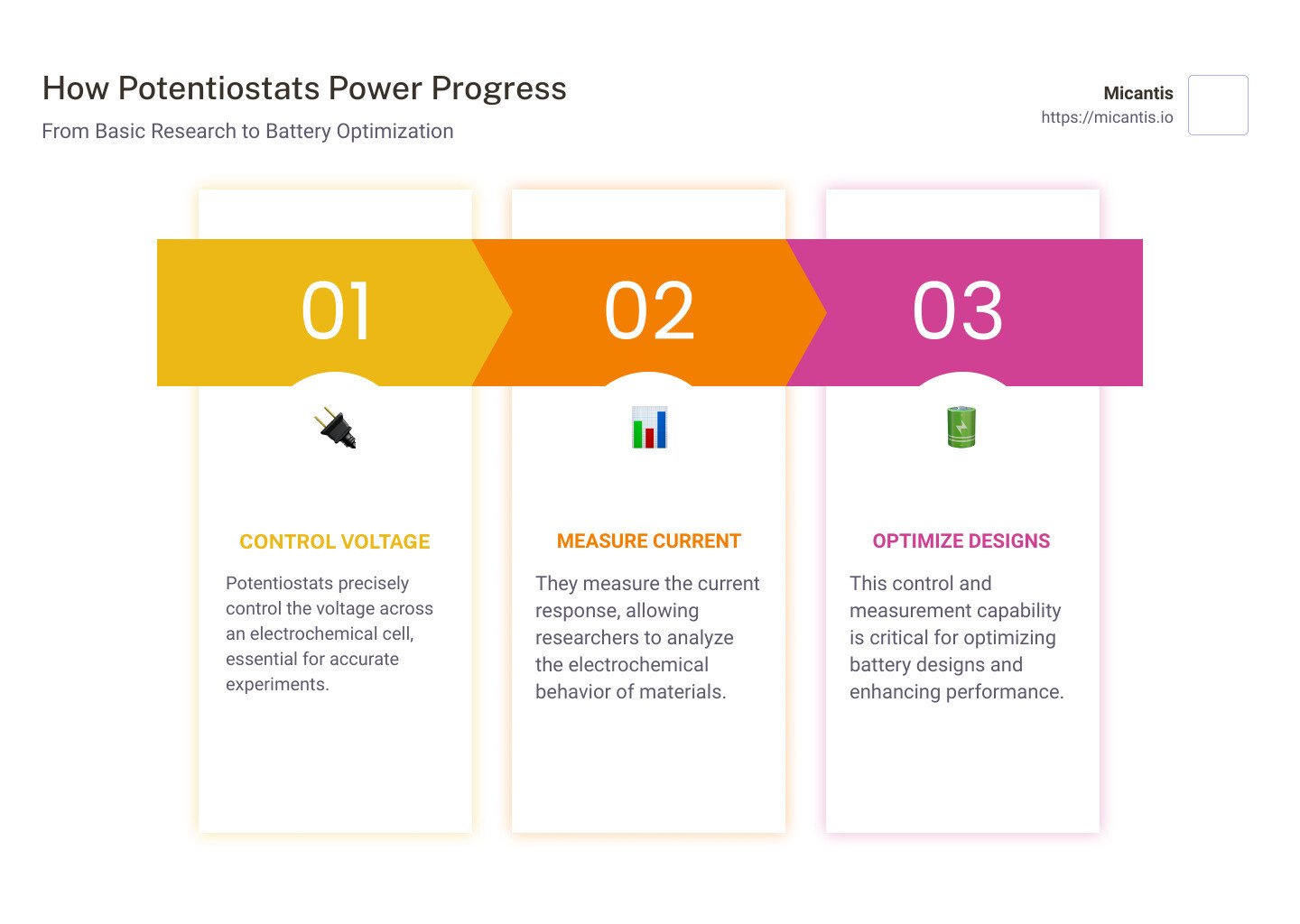
In short, a potentiostat:
- Controls the voltage across an electrochemical cell.
- Measures the current response due to this controlled voltage.
- Supports a wide range of electrochemical techniques, critical in fields from biochemistry to battery optimization.
Whether you're a battery designer grappling with the challenges of slow R&D cycles and high development costs, or an engineer at an EV manufacturer focused on quality testing, a deeper understanding of potentiostats could be the key to streamlining your processes.
What is a Potentiostat?
Imagine you're a scientist in a lab, looking to unlock the secrets of chemical reactions, or maybe you're an engineer trying to create the next generation of batteries. There's one tool that's going to be your best friend on this journey: the potentiostat.
What's a Potentiostat? In simple terms, a potentiostat is like a very sophisticated remote control for electrochemical experiments. It's a piece of electronic hardware that allows scientists to precisely control and measure the voltage and current in an electrochemical cell.
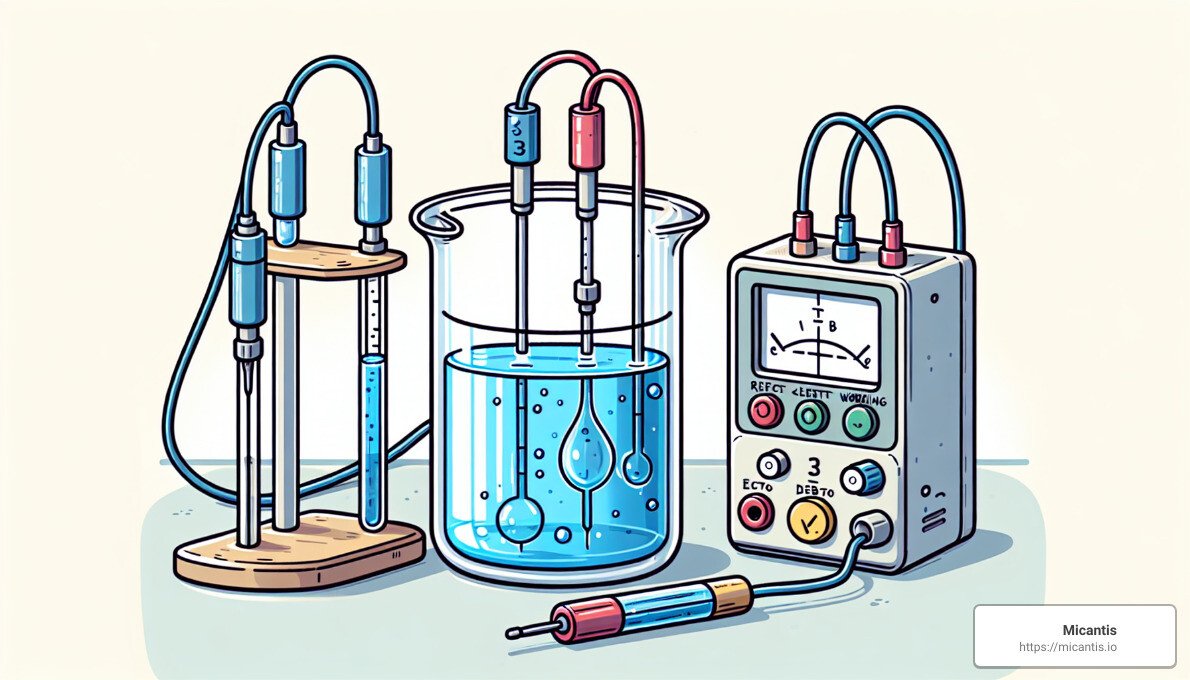
The Three-Electrode Cell: At the heart of these experiments is something called a three-electrode cell. This setup includes:
- The Working Electrode (WE): This is where the magic happens—chemical reactions are either started or stopped here.
- The Reference Electrode (RE): This is the gold standard electrode that helps keep the working electrode's voltage steady.
- The Counter (Auxiliary) Electrode (CE): This electrode completes the circuit, allowing the electricity to flow.
How Does a Potentiostat Work?
When we talk about a potentiostat, we're diving into the heart of electrochemistry. But how does this crucial piece of equipment operate?
Negative Feedback System
Imagine you're riding a bike up a hill, and you want to maintain a steady speed. If you start to slow down, you pedal harder. If you're going too fast, you ease off. This is similar to how the negative feedback system in a potentiostat works.Electrode Potential Control
Controlling the electrode potential is like setting the exact temperature you want on your air conditioner. The potentiostat applies a specific voltage between the working electrode and the reference electrode.Current Measurement
The potentiostat measures the flow of electric current between the working and counter electrodes as the reaction proceeds. This flow of electrons gives us valuable information about the electrochemical reaction.Applications of Potentiostats
Corrosion Research
Potentiostats can simulate different environments to see how metals react and help us understand why they corrode.Chemical Synthesis
Potentiostats help us understand the recipe - the step-by-step process of a chemical reaction.Battery Testing
Potentiostats act as coaches for batteries, testing their limits to see how much energy they can store and how many times they can be recharged.Analytical Chemistry
Potentiostats help us measure the tiny currents that flow as a result of chemical reactions.Conclusion
In the intricate world of electrochemistry, potentiostats play a pivotal role. At Micantis, we understand the hurdles researchers face—from data management to optimizing battery performance. Our platform and potentiostat technology are designed to make your research as straightforward and impactful as possible.
Join us at Micantis, and let's write the next chapter of electrochemical research together.